Newsletter 2025/1
msds-europe – newsletter 2025/1
MSDS-Europe newsletter
– January 2025 –
Current news

Regulation (EU) 2024/2865 unexpectedly entered into force on December 9, 2024
Significant legislative changes have been implemented in the field of chemical safety. The Regulation (EU) 2024/2865, which came into effect on December 9, 2024, introduces substantial modifications in the classification, labeling, and online sales regulations of certain substances and mixtures.
The new requirements primarily affect manufacturers, importers, and distributors, but they are also relevant for any company that uses or stores hazardous substances or mixtures. Below, we outline the key information and necessary actions.
Key changes introduced by Regulation (EU) 2024/2865
1. Designation of an EU-based supplier
For hazardous substances and mixtures originating from outside the EU it is mandatory to designate an EU-based supplier responsible for ensuring the legal compliance of the products.
This rule is particularly important for online sales, where consumers can order directly from manufacturers.
Effective date: 9 December 2024
Action required: Verify that all products originating from outside the EU have a designated EU-based supplier.
2. Online access to labelling information
When sold online, all hazardous substances and mixtures must clearly and prominently display the labelling elements of Article 17 of Regulation (EC) No 1272/2008 to the customers.
This rule ensures that customers have detailed information about the product before purchasing.
Effective date: 9 December 2024
Action required: Review your online sales platforms and ensure that labelling information is accessible for all products.
3. New classification and labeling requirements
The new regulations introduce further changes to the classification, labelling, and packaging of hazardous substances and mixtures:
- Introduction of new hazard classes:
Certain substances and mixtures now must be classified into new categories, such as:
- Endocrine disruptors (for human health and the environment).
- Persistent, bioaccumulative, and toxic (PBT) substances, as well as very persistent and very bioaccumulative (vPvB) substances.
- Updates to labels and packaging:
The regulations introduce new minimum font sizes and spacing to improve the readability of labels. For example:
- Packaging up to 0.5 liters: 1.2 mm
- 0.5–3 liters: 1.4 mm
- 3–50 liters: 1.8 mm
- 50–500 liters: 2.0 mm
To enhance readability, additional requirements include:
- Text must be printed in black on a white background.
- A uniform sans-serif font must be used.
- Line spacing must be at least 120% of the font size.
The regulation also allows for digital labelling and the use of fold-out labels, particularly for smaller packaging.
4. Rules for supply via refill stations
In practice, an increasing number of hazardous chemical products (e.g., detergents and cleaning agents) are sold without packaging. Specific rules and conditions have been introduced for such sales.
The CLP Regulation now includes definitions for refilling and refill stations (Article 2), while the detailed rules are provided in Article 35 (2a) and Annex II, Section 3.4.
The following hazard classes are prohibited from being distributed through refill systems:
- Acute toxicity (any category)
- Specific target organ toxicity – single and repeated exposure, any category
- Skin corrosion category 1, any subcategory
- Serious eye damage category 1
- Respiratory sensitisation, any category
- Skin sensitisation, any category
- Aspiration hazard
- Germ cell mutagenicity, any category
- Carcinogenicity, any category
- Reproductive toxicity, any category
- Flammable gases, any category
- Flammable liquids, categories 1 and 2
- Flammable solids, any category
- Endocrine disruptors for human health and the environment, any category
- Persistent, bioaccumulative, and toxic
- Very peristent and very bioaccumulative
- Peristent, mobile and toxic
- Very persistent and very mobile
5. PCN notification obligations
Please note that the transition period for hazardous mixtures ends on 31 December 2024.
Notifications submitted under previous national regulations will become “archived” after this date.
Starting in 2025 only PCN notifications compliant with Annex VIII of the CLP Regulation will be valid.
Action required: If not already done, ensure that PCN notifications are submitted for the affected mixtures as soon as possible.
Do you have any further questions?
Customer service Ask our chemical safety expert
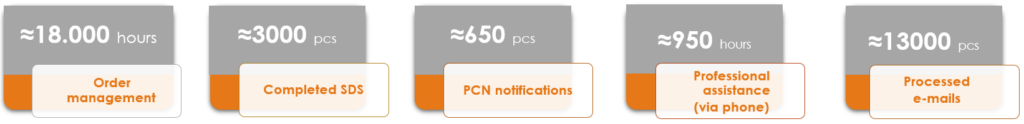
2024 in numbers

Wishing you a prosperous and successful 2025!
We thank our clients for their trust and our team for their dedicated work!
Pondering

SVHC: Substances of very high concern. Should we concern?
Candidate List of substances of very high concern (published in accordance with Article 59(10) of REACH) has been extended again:
07 November 2024 –Triphenyl phosphate (CAS No 115-86-6)
Under the REACH Regulation, restrictions have been introduced for certain Substances of Very High Concern.
These substances can only be used for purposes specified in Annex XIV of the REACH Regulation. Below is a summary of what these obligations might mean for stakeholders in practice.
In cases where it is impossible to replace a substance with a less hazardous alternative due to specific technological requirements, the use of such a substance may still be permitted under proper justification.
This would require authorization by the manufacturer, importer, or downstream user for uses not covered in Annex XIV.
Obligations related to articles and SVHCs
Information obligations
If the concentration of an SVHC in an article exceeds 0.1% by weight (at the component level in complex objects), businesses are required to inform their customers (excluding consumers). This information must include at least the substance’s name (preferably with a CAS number) and if applicable guidance on its safe use.
Notification obligations
Companies must submit a notification to the ECHA if:
- the SVHC concentration exceeds 0.1% by weight in articles and
- the total annual volume of the SVHC in the articles exceeds one tonne per producer or importer.
The notification must be submitted within six months of the SVHC being added to the Candidate List. However, notification is not required if:
- exposure to humans or the environment can be excluded during use and disposal, or
- the substance has already been registered for the specific use.
These obligations particularly affect companies dealing with articles, as practical experience shows that awareness of these requirements is often lacking in the industry.
Updates to Safety Data Sheets (SDSs)
Existing SDSs must be updated to reflect the identification of substances as SVHCs. For example:
- Must be revised to include the SVHC classification.
- SDSs must be provided even for non-hazardous mixtures if they contain at least 0.1% of an SVHC that is classified as PBT, vPvB, or of equivalent concern.
If you need help: Revision, update of SDS according to current regulations
What is the difference between the Candidate List and the Authorization List?
Candidate List:
The Candidate List contains substances identified as SVHCs by the European Chemicals Agency (ECHA).
- Purpose: To disclose potential risks and prepare for possible inclusion in the Authorization List.
- Obligation: Manufacturers, importers, and distributors must inform their customers about the presence of an SVHC if the concentration exceeds 0.1% by weight.
Authorization List (Annex XIV):
Substances on the Authorization List can only be used in the EU with explicit authorization.
- Purpose: To encourage the transition to safer alternatives and phase out particularly hazardous substances wherever possible.
- Obligation: Companies must apply for authorization from the ECHA to use substances on this list beyond the specified sunset date.
Regulatory change observer

COMMISSION DELEGATED REGULATION (EU) 2024/3199 of 15 October 2024
amending Regulation (EU) No 649/2012 of the European Parliament and of the Council as regards the listing of pesticides and industrial chemicals
Effective Date: January 20, 2025
This regulation must be applied starting from March 1, 2025.
Key Updates:
- Several active substances, including benfluralin, benthiavalicarb, clofentezine, dimethomorph, and S-metolachlor, did not receive approval renewals and are now banned in the “pesticides” category.
- Approvals for ipconazole and asulam-sodium were withdrawn, leading to a ban in pesticide applications.
- Industry withdrawals for substances like calcium phosphide, chloridazon, flusilazole, and oxadiazon have resulted in their inclusion in Annex I of Regulation (EU) No 649/2012, with bans for all uses.
- The harmonized classification of these substances highlights risks to human health and the environment, justifying their prohibition.
The updated Annex I significantly impacts the import and export of hazardous substances within the EU, requiring businesses to review and adjust their compliance strategies.
Regulatory change observer in the main EU languages
EN I DE I FR I ES I IT I PL I CS I RO I SL I SK
Do you have a question regarding the newsletter content? Ask our expert!

